On March 19, the international academic journal Nature published the full clinical research data of CNBG-Virogin's oncolytic virus VG161. The study was conducted on patients with advanced, refractory hepatocellular carcinoma (HCC) who had exhausted multiple lines of therapy. The results showed that VG161 exhibited not only exceptional safety but also proved statistically significant in prolonging patient survival, thereby providing a novel therapeutic option for those with advanced HCC.
The research data had previously garnered significant attention at the 2024 American Society of Clinical Oncology (ASCO) Annual Meeting and received the designation of a breakthrough therapy in China in September 2024. The publication in Nature further validates its scientific value and clinical potential, effectively enriching the evidence-based medical data.


The corresponding author of the article and principal investigator of the clinical trial is Professor Liang Tingbo, a hepatobiliary and pancreatic surgery expert at the First Affiliated Hospital of Zhejiang University School of Medicine. The first author and assistant investigator is Dr. Shen Yinan, a specially appointed researcher at the First Affiliated Hospital of Zhejiang University School of Medicine.
Key Data Highlights Clinical Benefits: Positive Efficacy and Excellent Safety
Significant Efficacy and Prolonged Survival
Participant Population: The study included 40 patients with advanced HCC who had failed multiple lines of standard treatment (including targeted therapy and immune checkpoint inhibitors). Among them, 92.5% had received ≥2 lines of treatment, and 85% had undergone immune checkpoint inhibitor therapy for over 3 months.
Objective Response and Disease Control: Among the 37 patients with evaluable efficacy, the objective response rate (ORR) was 18.92%, and the disease control rate (DCR) was 64.86%.
Survival Advantage: The median overall survival (mOS) for the 37 patients in the VG161-treated group was 12.4 months, representing a significant improvement compared to the survival outcomes observed with traditional second-line treatments. Notably, in a subgroup of 22 patients who had failed over 3 months of immunotherapy (PreCPI >3 months), the median OS reached 17.3 months.
Excellent Safety and Feasibility for Long-Term Treatment
No dose-limiting toxicity (DLT) was observed, and the maximum tolerated dose (MTD) was not reached during the dose escalation phase. These findings underscore the excellent safety profile of VG161, supporting its feasibility for long-term treatment.
The most common adverse reactions were fever (86.4%) and transient peripheral blood lymphopenia (84.1%).
Currently, there is a lack of standard treatment options for end-stage advanced HCC patients, and this critical disease area remains in dire need of effective therapies.
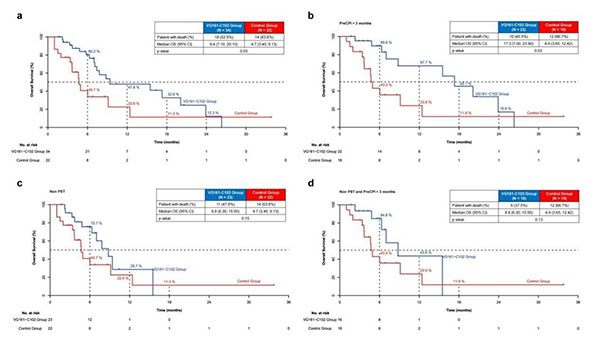
Figure a: The OS of the VG161 treatment group was significantly better than that of the real-world control group.
Figure b: The OS of the PreCPI >3 months treatment subgroup was significantly better than that of the ≤3 months treatment control group.
The study's findings indicated that, as a third-line therapeutic option for liver cancer, VG161 showed a median overall survival (OS) of 9.4 months, with efficacy comparable to that of standard second-line treatments.
Address: Sinopharm Plaza, No 20 Zhichun Road, Haidian district, Beijing
Postcode: 100191
Tel: 86-10-82287727
Fax: 86-10-62033332
- Sinopharm Group Co Ltd (Sinopharm Holding)
- China National Biotec Group Company Ltd
- China State Institute of Pharmaceutical Industry
- China Sinopharm International Corporation
- Reed Sinopharm Exhibitions Co Ltd
- China Traditional Chinese Medicine Holdings Co Ltd
- Shanghai Shyndec Pharmaceutical Co Ltd
- Sino Pharmengin Corporation
- China National Pharmaceutical Investment Co Ltd
- Sinopharm Group Finance Co Ltd
- China National Medicines Corporation Ltd
- China National Accord Medicines Corporation Ltd
- Beijing Tiantan Biological Products Co Ltd
- Chongqing Taiji Industry (Group) Co Ltd
- China Pharmaceutical Industry Association
- China National Narcotic Drugs Association
- China Association of Pharmaceutical Commerce
- China Pharmaceutical Innovation and Research Development Association
- China Association of Medicine Devices Industry
- China Association of Traditional Chinese Medicine
- China Pharmaceutical Culture Society




